 Creating these holes can be done with the point of a pencil or pen. We found that pre-punching a small hole in each element square on the periodic table makes insertion of the spaghetti pieces, done later, much easier. Now that your dough is ready, you'll need copies of a periodic table. The dough will end up being about 1/2 inch thick. Keeping it on the wax paper can make clean-up easier. Then flatten it into a rectangle about the size of an 8.5 x 11 inch sheet of paper. Once the dough is prepared and cooled, knead it on wax paper into a soft mass. We used food coloring but koolaid works, too. Coloring the dough is not necessary, but doing so can make it more fun or useful for other activities. Caution should be taken not to get burned. Either way you make it, microwave or stovetop, the dough will be hot once it forms into a ball. The key to success is the necessary stirring as the mixture heats. This same recipe can also be made on a stovetop surface. Here is a link to a microwave method we used which worked very well: There are many available recipes for making homemade playdough. We used 1 batch of dough per student, but this could be done in groups or as a demonstration. The making of the playdough can be done by your students or by you before your class begins. Instead of making a cake to build your 3-dimensional graph of the atomic radii of elements 1-49, we suggest you make a base of playdough. uncooked spaghetti noodles (a one-pound box is enough for 20+ students) copy of periodic table made on regular copy paper batch of homemade playdough (link to recipe is below) Whittaker EJW, Muntus R (1970) Ionic radii for use in geochemistry.Alternative Presentation for Radius Cake Activity found in Lesson _, page _ of Teacher's EditionĪlternative Presentation for Radius Cake Activity found in Lesson 10 Comment Phys-Math Soc Sci Fenn 1(38):1–25 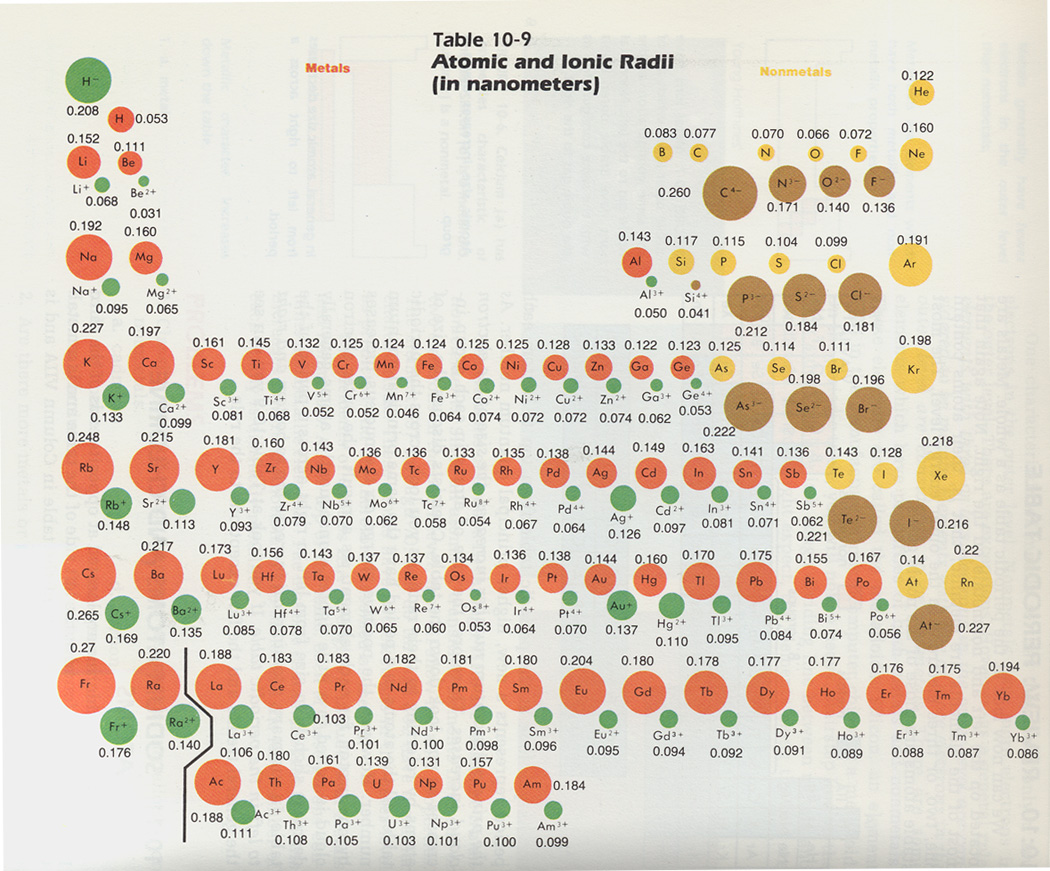
Wasastjerna JA (1923) On the radii of ions. Van Orman JA, Grove TL, Shimizu N (2001) Rare earth element diffusion in diopside: influence of temperature, pressure and ionic radius, and an elastic model for diffusion in silicates. Tiepolo M, Vannucci R, Oberti R, Foley SF, Botazzi P, Zanetti A (2000) Nb and Ta incorporation and fractionation in titanian pargasite and kaersutite: crystal-chemical constraints and implications for natural systems. Shannon RD, Prewitt CT (1969) Effective ionic radii in oxides and fluorides. 
In: O’Keeffe M, Navrotsky A (eds) Structure and bonding in crystals, vol II. Shannon RD (1981) Bond distances in sulphides and a preliminary table of sulfide crystal radii. Shannon RD (1976) Revised ionic radii and systematic studies of interatomic distances in halides and chalcogenides. /PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
Pauling L (1960) The nature of the chemical bond and the structure of molecules and crystals: an introduction to modern structural chemistry, vol 18. Landé CA (1920) Uber die Grösse der Atome. 
Goldschmidt VM (1926) Die gesetze der krystallochemie. Ghosh DC, Biswas R (2003) Theoretical calculation of absolute radii of atoms and ions. Am Mineral 74:620–626īlundy J, Wood B (2003) Partitioning of trace elements between crystals and melts. Bartelmehs KL, Gibbs GV, Boisen MB Jr (1989) Bond length and bonded-radii variations in sulfide molecules and crystals containing main-group elements: a comparison with oxides. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
